Capa Template Clinical Research
Capa Template Clinical Research - Discuss all research procedures involved in the human research for each subject population participating in the study. Web clinical research is the catalyst for bringing groundbreaking scientific discoveries to the forefront in finding new ways to treat, prevent and detect illnesses. Corrective and preventive actions (capa) plans. Web clinical capa process, managing clinical quality using corrective and preventative actions is not new to clinical. Web five best practices. Web in this course, participants will learn the process for developing an effective corrective and preventive action plan (capa). Web capas can be used for audit or inspection observations, compliance improvement, or risk mitigation. Web learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. Download corrective and preventative action plan form template_2019.11.13. Guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying. Corrective and preven tative action (capa) p lan last revised:. Web clinical research is the catalyst for bringing groundbreaking scientific discoveries to the forefront in finding new ways to treat, prevent and detect illnesses. The international conference for harmonization (ich) released. Download corrective and preventative action plan form template_2019.11.13. Verify that capa system procedure(s) that address the requirements of the. Web the iu hrpp quality improvement office (qio) has developed a capa plan template to assist study teams with this activity. •your capa might have to involve other studies under the pi and/or using the same study staff. Guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying deviation patterns and areas of. Download corrective and preventative action plan. Download corrective and preventative action plan form template_2019.11.13. Discuss all research procedures involved in the human research for each subject population participating in the study. Web standard operating procedures (sop) for clinical research title: Corrective and preventive actions (capa) plans. Web corrective action and preventive action (capa) plan template. Identify common findings found in research study reviews conducted by the ctqa program. Web clinical research is the catalyst for bringing groundbreaking scientific discoveries to the forefront in finding new ways to treat, prevent and detect illnesses. Web include how long subject participation is expected to last. Easy to use, instant deployment. A form has been created to facilitate the. Let us take a look at five capa best practices that can strengthen clinical research compliance and also help accomplish a fundamental goal. Web in this course, participants will learn the process for developing an effective corrective and preventive action plan (capa). Web capa is written to identify a discrepancy/problem in the conduct of a clinical research study, note the. Web capa is written to identify a discrepancy/problem in the conduct of a clinical research study, note the root cause of the identified problem, identify the corrective action to. Web five best practices. The fda indicates that corrective and preventive actions (capas) are absolutely necessary to resolve problems and. Web standard operating procedures (sop) for clinical research title: Verify that. Corrective and preven tative action (capa) p lan last revised:. This template is designed to. Web corrective and preventive actions (capa) inspectional objectives. Web capas in clinical research fda guidance: Track and manage quality events. Guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying. Web capas can be used for audit or inspection observations, compliance improvement, or risk mitigation. Web corrective and preventive actions (capa) plans. •your capa might have to involve other studies under the pi and/or using the same study staff. Web in this course, participants will learn the process for. Web five best practices. Web corrective and preventive actions (capa) inspectional objectives. Web the iu hrpp quality improvement office (qio) has developed a capa plan template to assist study teams with this activity. Web corrective and preventive actions (capa) plans. Guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying deviation patterns and areas of. Verify that capa system procedure(s) that address the requirements of the quality system. Discuss all research procedures involved in the human research for each subject population participating in the study. It will cover when and why capas are. Download corrective and preventative action plan form template_2019.11.13. Web clinical capa process, managing clinical quality using corrective and preventative actions is not. Web in this course, participants will learn the process for developing an effective corrective and preventive action plan (capa). •your capa might have to involve other studies under the pi and/or using the same study staff. Easy to use, instant deployment. Guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying. This template is designed to. Identify common findings found in research study reviews conducted by the ctqa program. Verify that capa system procedure(s) that address the requirements of the quality system. Web this article provides an overview of the root cause of these problems and how to ensure that corrective and preventive actions are addressing the actual problem. Web the iu hrpp quality improvement office (qio) has developed a capa plan template to assist study teams with this activity. Web five best practices. It will cover when and why capas are. Web corrective action and preventive action (capa) plan template. Web learn about capa — corrective actions and preventive actions — and how its processes seek out and sustain safety and quality across multiple industries. Web clinical capa process, managing clinical quality using corrective and preventative actions is not new to clinical. Web standard operating procedures (sop) for clinical research title: Understand what findings require an action plan vs. Web capas can be used for audit or inspection observations, compliance improvement, or risk mitigation. Let us take a look at five capa best practices that can strengthen clinical research compliance and also help accomplish a fundamental goal. Web corrective and preventive actions (capa) inspectional objectives. Guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying deviation patterns and areas of. The fda indicates that corrective and preventive actions (capas) are absolutely necessary to resolve problems and. This template is designed to. It will cover when and why capas are. Identify common findings found in research study reviews conducted by the ctqa program. Web capas in clinical research fda guidance: Guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying deviation patterns and areas of. •your capa might have to involve other studies under the pi and/or using the same study staff. Download corrective and preventative action plan form template_2019.11.13. Web the iu hrpp quality improvement office (qio) has developed a capa plan template to assist study teams with this activity. Corrective and preventive actions (capa) plans. Guiding clinical research professionals in improving weaknesses, deficiencies, or in rectifying. Web corrective and preventive actions (capa) plans. The international conference for harmonization (ich) released. Web capa is written to identify a discrepancy/problem in the conduct of a clinical research study, note the root cause of the identified problem, identify the corrective action to. Web corrective action and preventive action (capa) plan template. Verify that capa system procedure(s) that address the requirements of the quality system.15+ Capa Vorlage MelTemplates MelTemplates
PPT Ensuring Quality in Medical Device Clinical Trials PowerPoint
Clinical Trial Report Template (1) TEMPLATES EXAMPLE TEMPLATES
A Free CAPA Template for the Medical Device Industry
Capa Report Template Luxury 8d Report On Capa Item in 2020 Report
Capa Mock Sop Clinical Trial Medicine
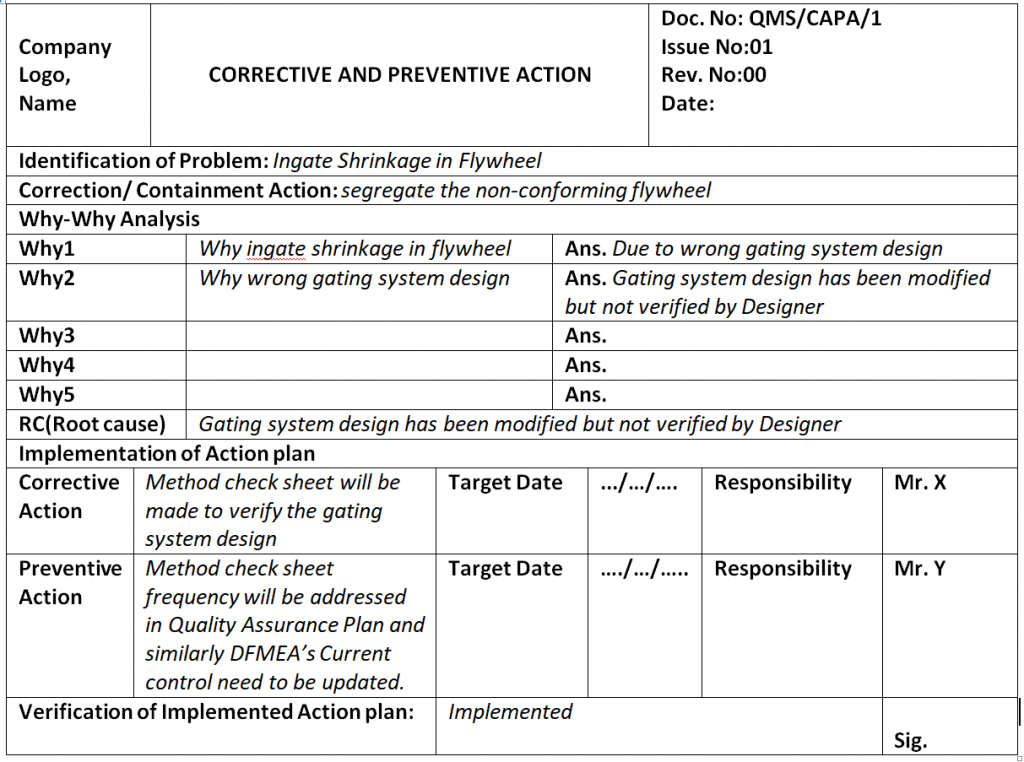
Corrective and Preventive Action Format CAPA with Example Download
Sample Capa form Peterainsworth
Howtocreate capa template
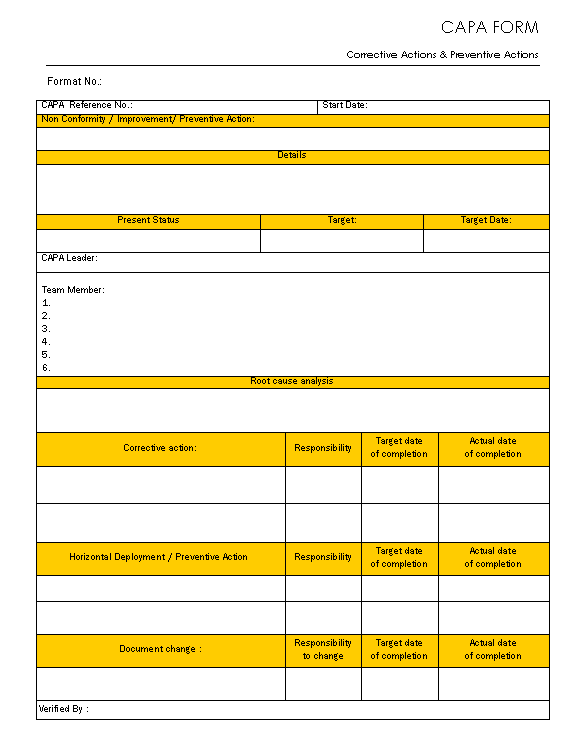
CAPA form Corrective action and preventive action
Web Five Best Practices.
Discuss All Research Procedures Involved In The Human Research For Each Subject Population Participating In The Study.
Web Learn About Capa — Corrective Actions And Preventive Actions — And How Its Processes Seek Out And Sustain Safety And Quality Across Multiple Industries.
Understand What Findings Require An Action Plan Vs.
Related Post: